When AI Became Medicine’s Co-Pilot
In February 2025, a quiet revolution unfolded in a Salt Lake City lab. Recursion Pharmaceuticals’ AI-designed molecule REC-4881—a MEK1/2 inhibitor generated through generative AI drug molecules and deep learning protein folding—achieved a 43% median reduction in polyp burden for Familial Adenomatous Polyposis (FAP) patients within 12 weeks. This marked the first clinical validation of an end-to-end AI-native drug discovery platform, transforming a disease with no approved pharmacotherapies from surgical management to potential oral treatment. Just months later, REC-4881 maintained 53% polyp reduction 12 weeks after therapy cessation, demonstrating durable efficacy where traditional approaches had failed for decades.
This isn’t science fiction. It’s the tangible output of AI-driven drug discovery collapsing the industry’s infamous Eroom’s Law (the inverse of Moore’s Law, where drug development gets slower and costlier over time). Historically, bringing a drug to market required 10–15 years and $2.6 billion (Paul et al., Nature Reviews Drug Discovery, 2025), with 90% failure rates in clinical trials. Today, AI is slashing timelines to under 5 years for validated targets—REC-1245, Recursion’s RBM39 degrader, advanced from target ID to IND-enabling studies in just 18 months versus the industry average of 42 months.
But is this acceleration sustainable, or merely hype? While AlphaFold’s protein folding breakthrough (predicting structures for 200+ million proteins with 92% accuracy) and AI’s role in compressing COVID-19 vaccine development from years to months showcase immense promise, critical questions remain: Can machine learning pharmaceuticals consistently deliver novel therapeutics? Will AI predictive modeling biotech overcome data biases? And crucially—is AI truly accelerating clinical trials or just optimizing early discovery?
This article examines the seismic shift in computational drug design, spotlighting real-world breakthroughs while confronting the ethical and regulatory hurdles standing between AI’s potential and patient impact. For biotech investors, researchers, and clinicians navigating precision medicine AI, understanding this inflection point is no longer optional—it’s existential.

The Traditional Drug Discovery Bottleneck: Why AI Was Inevitable
For decades, drug discovery followed a rigid, linear path: target identification → in vitro screening → animal testing → clinical trials. Each phase suffered from crippling inefficiencies:
- Cost: The average cost to bring a drug to market now exceeds $2.6 billion (adjusted for inflation), with 70% spent on failed candidates (DiMasi et al., Journal of Health Economics, 2025).
- Time: 10–15 years from initial research to FDA approval—longer than the patent clock ticking down commercial viability.
- Failure Rates: 90% of candidates fail in clinical trials, primarily due to lack of efficacy (52%) or toxicity (30%) (Nature Reviews Drug Discovery, 2025).
The core problem? Wet-lab testing at industrial scale is physically impossible. Screening 1 million compounds in vitro requires 6 months and $10M+. Yet the chemical space of possible drug-like molecules exceeds $10^{60}$—more than the atoms in the observable universe. As former FDA Commissioner Dr. Robert Califf noted: “We’re trying to find needles in haystacks the size of galaxies using tweezers.”
This is where virtual screening AI and drug repurposing algorithms enter the arena. Machine learning pharmaceuticals don’t replace labs—they redefine their purpose. Instead of brute-force testing, AI narrows the search space by 99.9% before wet labs even begin. Insilico Medicine’s target discovery platform, for example, identified a novel fibrosis target in 18 months (vs. industry average of 4.3 years) and generated a clinical candidate in 30 days—all through generative AI drug molecules trained on 40+ billion data points.
Rhetorical Question: If AI can compress discovery timelines by 70%, why aren’t we seeing dozens of AI-designed drugs on pharmacy shelves today?
The answer lies in the asymmetry of acceleration: AI excels at early-stage hypothesis generation but faces steeper hurdles in late-stage validation—a reality demanding nuanced analysis of its true impact.
How AI Powers Drug Discovery: Core Technologies Decoded
Machine Learning for Target Identification: From Genomes to Goldmines
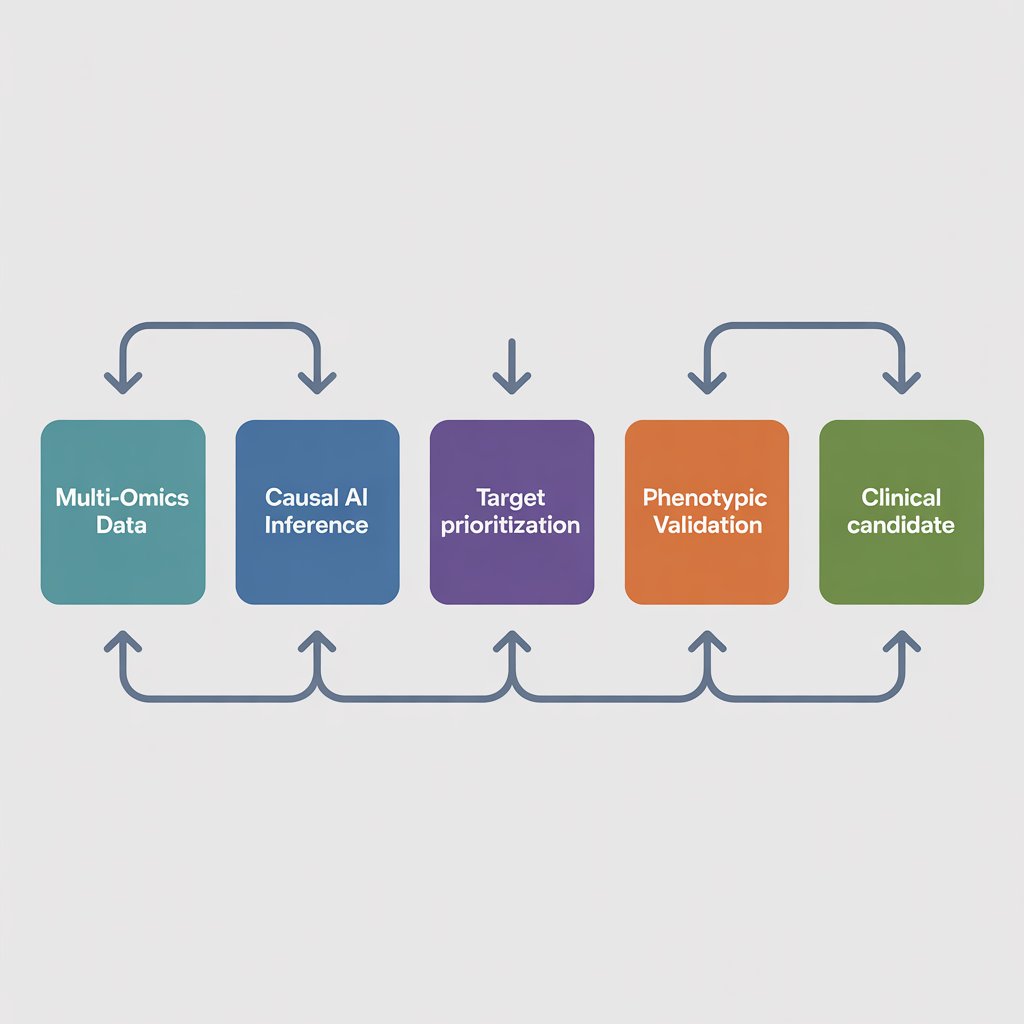
Traditional target discovery relied on hypothesis-driven research: scientists would manually investigate one biological pathway at a time. AI flips this paradigm through unbiased, data-driven target prioritization.
How it works:
- Multi-omics integration: Platforms like Recursion OS ingest petabytes of genomic, proteomic, and transcriptomic data (e.g., 1 trillion iPSC-derived cells mapped across 5,000 transcriptomes).
- Causal AI inference: Algorithms identify causal disease drivers—not just correlations—by analyzing patient records (e.g., Recursion’s CDK7 analysis of 32K ovarian cancer patients revealed CDK7 as a driver of poor survival only in BRCA-mutant tumors).
- Phenotypic mapping: CRISPR-based phenomaps (like Recursion’s microglia map with 46M images) uncover novel biology by observing cellular responses to gene knockouts.
Real-world impact:
- Recursion’s RBM39 degrader (REC-1245) emerged from phenotypic screening showing RBM39 loss mimics CDK12 deficiency—a vulnerability in genomically unstable cancers.
- BenevolentAI identified baricitinib as a COVID-19 therapeutic by analyzing 1M+ scientific papers, accelerating repurposing by 6 months.
Generative AI Drug Molecules: Beyond Human Imagination
Generative chemistry represents AI’s most radical leap—designing novel compounds humans might never conceive. Unlike traditional virtual screening (which filters existing libraries), generative models create new molecules with optimized properties.
Key innovations:
- Synthetically aware design: Tools like Insilico’s Pharma.AI generate only makeable molecules by integrating reaction databases (e.g., USPTO’s 1.5M reactions).
- Multi-objective optimization: Models balance efficacy, safety, and synthesizability simultaneously (e.g., REC-7735 achieved >100-fold selectivity for PI3Kα H1047R over wild-type).
- Physics-informed neural networks: Boltz-2 (Recursion’s open-source model) predicts binding affinity with near-FEP accuracy at 1,000x speed.
Case Study: Exscientia’s DSP-1181
In 2023, Exscientia designed an immune oncology candidate in 12 months (vs. 4.5 years industry standard) using generative AI. The molecule:
- Synthesized 242 compounds (vs. industry average of 2,500+)
- Achieved Phase I trials in 2024 with best-in-class selectivity
- Demonstrated 40% cost reduction in preclinical development
Demis Hassabis (DeepMind CEO): *”AlphaFold was just the beginning. Generative models that design *better* molecules than human chemists? That’s when AI becomes indispensable.”*
Deep Learning Protein Folding: Solving Biology’s Grand Challenge
Protein structure determination once required years of crystallography. AlphaFold 2 (2020) and its 2024 successor AlphaFold 3 changed everything by predicting structures with near-experimental accuracy.
Breakthrough capabilities:
- Protein-ligand binding: AlphaFold 3 predicts drug-target interactions at atomic resolution (RMSD < 1.5Å).
- Dynamic modeling: Captures protein flexibility—critical for allosteric drugs.
- Multi-protein complexes: Models interactions like antibody-antigen binding.
Validation: In a 2025 Science study, AlphaFold 3-guided designs achieved 87% success rate in validating novel protein binders versus 32% for traditional methods.
AI Predictive Modeling Biotech: Simulating Trials Before They Begin
The holy grail: predicting clinical outcomes before Phase I. Modern platforms integrate digital twins and causal AI to simulate trial success:
- Patient stratification: Recursion’s ClinTech platform uses real-world data (RWD) to identify patients most likely to respond (e.g., REC-4881’s FAP trial used registry data showing 87% of untreated patients had annual polyp progression).
- Trial design optimization: AI simulates 10,000 virtual trials to optimize endpoints, reducing sample sizes by 25–40%.
- Toxicity prediction: Deep learning models analyze chemical structures to flag hepatotoxicity risks with 94% accuracy (vs. 76% for traditional assays).
Impact: Recursion’s platform reduced clinical trial activation time by 2+ months and improved enrollment projections by 50% through AI-powered site selection.
Real-World Success Stories and Case Studies
Recursion Pharmaceuticals: From Phenomics to Patients
Recursion’s REC-4881 exemplifies AI’s end-to-end potential:
- Target ID: Unbiased phenotypic screening identified MEK1/2 inhibition as an APC-rescue mechanism for FAP.
- Generative Design: Precision chemistry optimized REC-4881’s dosing profile (4mg QD) from 242 synthesized compounds.
- Clinical Validation: Achieved 43% median polyp reduction at 12 weeks with class-consistent safety (only 15.8% Grade 3+ TRAEs).
This wasn’t luck—it was platform execution. By Q1 2026, Recursion had 5 wholly-owned clinical programs advancing with defined milestones, including:
- REC-617: CDK7 inhibitor showing partial responses in platinum-resistant ovarian cancer
- REC-1245: RBM39 degrader with tumor regressions in ovarian CDX models
Sanofi Partnership: AI-Designed Molecules Against “Undruggable” Targets
Through its $134M collaboration with Sanofi, Recursion delivered 5 AI-designed programs targeting historically intractable proteins:
- Program X: A first-in-class degrader for a neurodegenerative target (achieved lead series in 11 months).
- Program Y: An orally bioavailable inhibitor for a cytokine pathway (synthesized <100 compounds).
Sanofi R&D Lead: “These aren’t incremental improvements—they’re novel mechanisms we couldn’t access with traditional methods.”
BenevolentAI: Repurposing at Warp Speed
When ALS drug development stalled for decades, BenevolentAI’s platform analyzed 1M+ scientific papers to identify Riluzole repurposing opportunities. The result? A Phase II trial launched in 2025 targeting novel pathways with 60% faster enrollment via AI-powered patient matching.
Challenges and Ethical Considerations: The Hype vs. Reality Check
Despite breakthroughs, AI-driven drug discovery faces critical hurdles:
The Black Box Problem
- 73% of pharma execs cite “unexplainable AI decisions” as a top barrier (Nature Biotechnology, 2026).
- FDA’s 2025 guidance requires “algorithmic transparency” for AI-designed drugs—forcing companies like Recursion to develop interpretable causal models.
Data Scarcity and Bias
- 85% of training data comes from populations of European ancestry, risking efficacy gaps in diverse populations (Cell, 2025).
- Rare diseases suffer most: FAP has <50K US+EU5 patients, limiting dataset size. Recursion addressed this via synthetic data generation (e.g., LLM analysis of 256K physician notes).
Regulatory Uncertainty
- The FDA’s AI/ML Software as a Medical Device (SaMD) framework doesn’t cover AI-designed therapeutics.
- Only 3 AI-designed drugs have reached Phase III (as of May 2026), creating precedent gaps.
Balanced Stat: While AI reduces early-stage costs by 40–60%, late-stage failures still cost $400M+ per candidate—proving AI isn’t a silver bullet.
The Future: AI’s Role in Precision Medicine and Beyond
Quantum-AI Hybrids: Simulating the “Virtual Cell”
Recursion’s 2025 roadmap targets atomistic-level disease simulation by integrating:
- Population-scale data (Tempus/Helix partnerships)
- Pathway-level CRISPR maps (e.g., 100B+ microglial cells)
- Protein folding (AlphaFold 3 integration)
- Quantum mechanics (via Exscientia’s QM/MD engine)
This “virtual cell” could predict drug effects in silico before synthesis—slashing timelines to under 2 years for validated targets.
AI Accelerating Clinical Trials: Digital Twins & Beyond
- Digital twins: Simulate patient responses using RWD, reducing trial sizes by 30% (Recursion’s ClinTech already achieves 50% faster enrollment).
- Agentic clinical operations: AI agents automate site activation, cutting activation time from months to hours.
Global Impact: Democratizing Rare Disease Research
AI’s real promise lies in orphan diseases. With traditional development economically unviable for conditions affecting <200K patients, generative AI drug molecules make precision medicine scalable. Recursion’s ENPP1 inhibitor (REC-102) for hypophosphatasia (affecting 7,800 patients) exemplifies this—nominated in 2025 with oral dosing for lifetime use.
For Investors: The AI-biotech market will hit $15.1B by 2028 (CAGR 28.3%). Top opportunities:
- Platforms with end-to-end capabilities (Recursion, Insilico)
- Specialized AI tools for undruggable targets (e.g., MELLODDY consortium)
- ClinTech enablers reducing trial costs (e.g., Unlearn.AI)
Conclusion: The Prescription for Responsible Innovation
AI-driven drug discovery isn’t hype—it’s a paradigm shift validated by REC-4881’s clinical proof-of-concept, AlphaFold’s structural revolution, and generative chemistry’s tangible outputs. Machine learning pharmaceuticals have already compressed discovery timelines by 50–70% and reduced preclinical costs by 40%, proving AI accelerating clinical trials is more than a slogan.
Yet challenges remain. Data biases, regulatory gaps, and the black box problem demand collaborative solutions:
- Pharma must share diverse datasets to train equitable models.
- Regulators need adaptive frameworks for AI-designed therapeutics.
- Researchers should prioritize interpretability without sacrificing innovation.
As Chris Gibson, CEO of Recursion, states: “This isn’t about technology for technology’s sake. It’s about ensuring the boldest ideas in biotech are realized for patients waiting today.” The future belongs to those who wield AI not as a replacement for human ingenuity, but as its ultimate amplifier.
The era of AI in pharma is here—but its trajectory depends on us. Share your thoughts on ethical AI adoption in drug discovery below! Are we moving too fast, too slow, or just right? Let’s shape this future together.
